| Quick Answer Food safe packaging materials are materials certified to have no harmful chemical migration into food as part of a broader customized packaging solution. The primary categories are: food-grade plastics (PET, HDPE, PP), aluminum, glass, paper and paperboard with food-safe coatings, and biodegradable bioplastics. In the US, these are regulated by FDA 21 CFR. In the EU, Regulation (EC) No 1935/2004 governs them. Any packaging that directly contacts food must carry material compliance documentation and must never leach substances above established migration limits. |
The Day a Packaging Choice Almost Ended a Brand
A small organic snack company in Austin, Texas, spent 14 months building their product line. Their granola bars were exceptional. Their branding was clean. Their retail buyers were excited.
Then a routine compliance audit flagged their inner packaging liner. The material they were using had no food contact compliance documentation. The retailer pulled the order. The brand lost $80,000 in committed purchase orders and spent four months and roughly $22,000 fixing their supply chain. All of it preventable.
That story is not unusual. What is unusual is how little most brand owners know about food safe packaging materials before they need to. This article fixes that. Whether you are launching a food product, reformulating your packaging, or trying to survive a compliance review, this is the resource you should have read first.
Read more: — Packaging Materials Guide: What Brands Use to Protect Products
What Actually Makes a Packaging Material Food Safe?
Most people assume food safe means non-toxic. That is necessary but not sufficient. A material earns food contact status by passing a series of migration tests that measure how much of any given substance moves from the packaging into food under real-world conditions.
Migration happens in two ways when evaluating materials used in medicine boxes. Global migration measures the total amount of substances moving from packaging to food. Specific migration measures individual chemicals of concern, like bisphenol A (BPA), phthalates, or heavy metals such as lead and cadmium.
Both the US FDA and the European Food Safety Authority (EFSA) set legal limits for both types. In the EU, the overall migration limit for food contact materials is 10 mg per square decimeter of packaging surface. For specific substances, limits can be far lower, sometimes in the parts-per-billion range.
| What Food Contact Compliance Requires1. The material must be produced from approved substances listed in regulatory inventories (FDA 21 CFR or EU Regulation 10/2011 for plastics). 2. Migration testing must occur under conditions matching the intended food type (aqueous, fatty, acidic, dry). 3. A Declaration of Compliance (DoC) must accompany the material through the supply chain. 4. Documentation must be available for audit within 72 hours in most markets. |
The Four Conditions That Change Migration Risk
Migration is not fixed. It changes based on four variables that every brand must factor into material selection.
• Contact temperature: Hot-fill products push more migration than ambient-fill products.
• Contact time: A food stored for 24 months extracts more than a food consumed within 7 days.
• Food type: Fatty foods extract lipophilic substances more aggressively than watery or dry foods.
• Surface area to food volume ratio: Smaller packaging with high food contact surface is higher risk.
A brand selling a single-serve, high-fat nut butter sachet for 18-month shelf life in supplement packaging faces a completely different compliance picture than a brand selling dry crackers in a paperboard carton. Understanding your specific migration profile is step one.
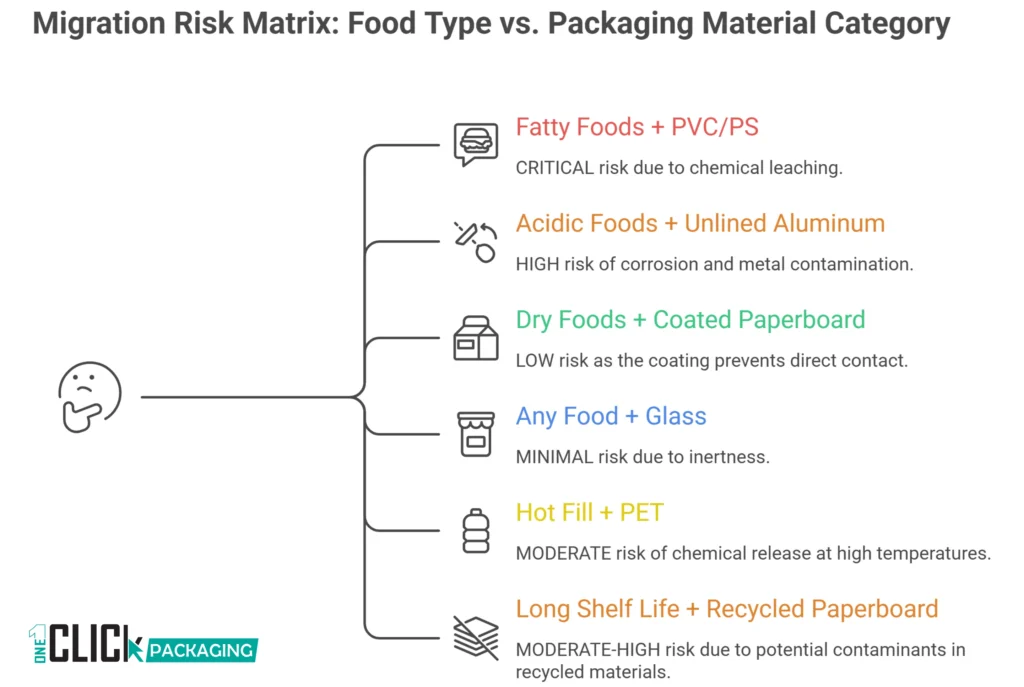
Food Safe Plastics: Which Ones Pass and Which Ones Should Concern You
Plastic accounts for roughly 40% of global food packaging boxes by volume. It is also the category where compliance complexity is highest and where brands get into the most trouble.
Here is a direct breakdown of the seven resin identification codes and their food contact status.
| Resin Code | Material | Food Safe Status | Common Food Use | Key Watch Point |
| #1 PET | Polyethylene Terephthalate | Yes — widely approved | Water bottles, salad containers, snack pouches | Not designed for reuse or high temps |
| #2 HDPE | High-Density Polyethylene | Yes — very safe | Milk jugs, juice bottles, cereal box liners | Avoid colored HDPE with unknown pigments |
| #3 PVC | Polyvinyl Chloride | Restricted — avoid | Cling wrap (fading out), deli containers | Phthalate plasticizers are a compliance risk |
| #4 LDPE | Low-Density Polyethylene | Yes — approved | Bread bags, squeeze bottles, some pouches | Confirm virgin-grade, not recycled |
| #5 PP | Polypropylene | Yes — excellent for hot fill | Yogurt cups, deli containers, microwave trays | Check additives and colorants separately |
| #6 PS | Polystyrene | Limited — context dependent | Foam trays, clear deli cups | Styrene monomer migration concern with fatty foods |
| #7 Other | PC, ABS, PLA, bio-blends | Varies — must verify each | Specialty containers, compostables | BPA in PC; PLA needs industrial composting disclosure |
The BPA Situation in 2026
Bisphenol A was removed from baby bottles across the EU and US years ago. But in 2024 and 2025, EFSA dramatically lowered its Tolerable Daily Intake for BPA from 4 micrograms per kilogram of body weight to 0.2 nanograms. That is a 20,000-fold reduction. Brands using BPA-containing can linings, jar lids, or polycarbonate components for any food product need current third-party testing to confirm compliance at the new threshold.
BPA-free does not automatically mean safe. Many replacements such as BPS and BPF are now facing their own scrutiny. Honest brands ask for migration data, not just marketing claims.
Paper and Paperboard: The Category That Surprises Most Brands
Paper used in custom printed food boxes feels inherently natural and safe. That intuition is mostly correct for uncoated papers. The complications arise from coatings, adhesives, printing inks, and recycled fiber content.
Recycled paperboard can contain mineral oils from newspaper printing inks. These mineral oil aromatic hydrocarbons (MOAH) and mineral oil saturated hydrocarbons (MOSH) migrate into Dry foods, especially cereals, pasta, and rice in coffee boxes and similar packaging, which lack a functional barrier. The European Food Safety Authority has flagged MOSH and MOAH migration as a priority concern since 2017, and Germany’s Federal Institute for Risk Assessment (BfR) has issued multiple warnings.
What Makes Paperboard Food Safe in Practice
• Virgin fiber paperboard eliminates the MOSH/MOAH risk from recycled content.
• Functional barrier layers (inner PE, PLA, or foil laminate) block migration from outer board layers.
• Food-safe printing inks must be used on any layer contacting food or migrating through barrier layers.
• Water-based and UV-cured inks have different migration profiles — confirm your printer’s compliance documentation.
• Grease-resistant paperboard often uses per- and polyfluoroalkyl substances (PFAS), which are increasingly restricted or banned.
PFAS in Food Packaging: A 2026 Priority Issue
PFAS chemicals were used widely in grease-resistant food packaging for pizza boxes, fast food wrappers, microwave popcorn bags, and bakery bags. Denmark banned them in food contact materials in 2020. The US FDA requested voluntary phase-outs in 2020 and 2021, with several major manufacturers complying. California’s AB 1200, effective January 2023, banned intentionally added PFAS in food packaging.
If your product line includes any grease-bearing food in paperboard packaging, confirming PFAS-free status from your supplier is now a basic compliance requirement in most developed markets. Ask specifically for third-party test results, not just a supplier declaration.
Glass and Aluminum: The Materials That Almost Never Fail Compliance
Glass used in jar packaging has been used for food contact for centuries and remains the most chemically inert food packaging material available commercially. It has no migration of concern, no endocrine-disrupting compounds, and no reaction with acidic, fatty, or aqueous foods. The compliance question with glass packaging is never the glass itself. It is the lid.
Metal closures on glass jars traditionally used plastisol sealants containing BPA. As of 2023, major closure manufacturers including Silgan and Pano Cap have transitioned to BPA-free compounds. Brands using glass packaging should confirm their closure supplier’s current compound formulation and request DoC documentation annually, because formulations change.
Aluminum: Cans, Foil, and Laminate
Aluminum is food safe when used correctly. The complication is the internal can lining. Until recently, most aluminum beverage and food cans used epoxy linings containing BPA. Following EFSA’s 2023 opinion and FDA pressure, the industry has been transitioning to acrylic, polyester, and olefin-based alternatives. As of early 2026, Ball Corporation and Crown Holdings have both announced completion of their BPA-free lining transitions for major product lines, though branded can lines from smaller manufacturers may still use legacy formulations.
Aluminum foil and laminate structures used in flexible packaging are generally compliant, but the adhesive layers in laminated structures require independent verification. Solvent-based adhesives can leave residues that migrate into food, particularly at elevated temperatures.

Bioplastics and Compostable Packaging: Promising, But Not Automatically Safe
This is where brands make the most optimistic assumptions and face the most unexpected compliance gaps. Bioplastic does not mean food safe by default. PLA (polylactic acid), PHA (polyhydroxyalkanoate), and starch-based blends must be tested for migration just like any other plastic.
PLA specifically is approved for food contact under FDA regulations and EU Regulation 10/2011, but only specific grades manufactured under controlled conditions. A PLA cup sourced without food contact certification is not automatically compliant, even though the base polymer is generally recognized as safe.
The Compostability Labeling Trap
Compostable packaging and food safe packaging are independent claims. A compostable material can be non-compliant for food contact. A food safe material may not be compostable. Brands pursuing sustainable packaging must verify both properties independently and document both separately.
In 2024, several natural food brands faced FTC scrutiny for compostability claims on packaging that failed to decompose in industrial composting facilities within the claimed timeframe. The FTC Green Guides now require specificity: industrial compostable, home compostable, or neither.
The Documentation Stack Every Brand Must Maintain
Understanding food safe materials is half the work. The other half is maintaining documentation that proves compliance. During a regulatory inspection, an FDA recall investigation, or a major retailer audit, documentation gaps are treated the same as material failures.
Five Documents Every Brand Needs in Their Packaging File
• Declaration of Compliance (DoC): Issued by your packaging supplier, confirming the material meets applicable regulations for your specific food type, temperature, and contact duration.
• Migration Test Reports: Third-party laboratory results showing migration testing under conditions matching your actual product. SG S, Intertek, and Eurofins are the three most commonly accepted testing laboratories globally.
• Substance Inventory Records: List of all substances used in the material above 0.01% (100 ppm) as required under EU Regulation 10/2011 for plastics.
• Supplier Qualification Records: Evidence that your packaging supplier operates a quality management system relevant to food contact materials (ISO 22000, FSSC 22000, or equivalent).
• Traceability Records: Lot-level traceability linking each production run of your product to the specific batch of packaging material used, enabling targeted recalls if needed.
How to Evaluate a Packaging Supplier’s Food Safety Claims
Supplier declarations are only as trustworthy as the testing behind them. Here is how to separate genuine compliance programs from paper compliance.
| Evaluation Question | Green Flag | Red Flag |
| Can you provide a current DoC specific to my food type? | Yes, issued within 12 months, specific to aqueous/fatty/dry food category | Generic DoC not specific to food category or older than 3 years |
| Who conducted your migration testing? | Accredited third-party lab (Eurofins, SGS, Intertek) | Internal testing only with no accreditation |
| Are your colorants and additives separately listed? | Yes, with individual substance compliance confirmation | Colorants covered under general DoC without detail |
| Do you hold any food contact certifications? | ISO 22000, FSSC 22000, or BRC packaging standard | No certification, only self-declaration |
| How do you handle formulation changes? | Written notification policy, updated DoC within 30 days | No formal process for notifying customers |
The Practical Takeaway for Brand Owners in 2026
The regulatory environment for food safe packaging materials is tightening, not loosening. PFAS restrictions are expanding state by state and country by country. The BPA tolerable intake revision has made testing thresholds stricter. Compostability claims are under scrutiny from the FTC. And retailer audits, particularly from Whole Foods, Costco, and Target, are requesting compliance documentation at a level they were not three years ago.
The brands that handle this well are not the ones with the largest compliance teams. They are the ones that started early, asked the right questions of their suppliers, and built documentation habits before they needed them under pressure.
Start with your highest-risk packaging, the materials in direct contact with your fattiest, most acidic, or longest-shelf-life products. Get current DoC documentation. Get a third-party migration test. Put both in a folder that any employee can find in ten minutes. Then do the same for every other packaging component, working down the risk list.
One more thing: your packaging supplier’s compliance program is only as strong as the questions you ask. Suppliers with nothing to hide welcome detailed compliance questions from brands working with a reliable packaging solutions provider. The ones who respond to compliance requests with vague reassurances are telling you something important.
Frequently Asked Questions (From Reddit, Quora, and Real Brand Pain Points)
Is food grade the same as food safe?
Not exactly. Food grade means the material is made from substances approved for food contact. Food safe means it has been tested and verified to not transfer harmful substances to food under your specific conditions of use. A material can be food grade but not food safe if it is used outside its intended application, such as using a room-temperature-rated material for hot-fill processing.
Does food safe packaging need FDA approval?
The FDA does not approve individual packaging materials in the way it approves drugs. Instead, it maintains lists of substances that are Generally Recognized As Safe (GRAS) or approved under Food Contact Notifications (FCNs) and 21 CFR regulations. Your packaging material must be made from substances on these lists. The compliance burden is on the manufacturer and brand, not a pre-market FDA sign-off process.
Can I use any plastic container for food storage?
No. Plastics not designed for food contact, such as general-purpose storage bins, industrial containers, or packaging not carrying food contact marking, can leach plasticizers, stabilizers, and processing aids into food. Always look for the food contact symbol (a fork and wine glass icon) or confirm food contact certification from the supplier before use in food applications.
What does the fork and glass symbol on packaging mean?
It means the material has been tested and confirmed suitable for food contact under EU regulations. It does not automatically appear on US packaging, which relies on regulatory substance lists rather than on-pack symbols. The symbol is a positive signal but does not replace reviewing the actual Declaration of Compliance, especially for international brands operating across markets.
How often should migration testing be repeated?
Testing should be repeated when your packaging supplier changes their formulation, when you change suppliers, when you add a new food type to a package used across multiple SKUs, when you change filling temperature or storage conditions, or at minimum every three to five years as regulatory standards evolve. EFSA’s 2023 BPA revision is a real-world example of why periodic re-testing matters even when nothing in your supply chain has changed.
Are Kraft paper bags food safe?
Unbleached Kraft paper used in kraft packaging boxes in direct food contact is generally considered safe for dry, non-fatty foods. The risk increases with wet or fatty foods, where MOSH/MOAH migration and potential microbial penetration become concerns. Many Kraft bags also use recycled fiber, which introduces greater uncertainty. For high-fat or moist foods, inner PE or PLA liners are standard practice.
What packaging is safest for acidic foods like tomato sauce or citrus?
Glass is the gold standard for acidic foods due to zero reactivity. Aluminum cans with properly certified epoxy or acrylic linings are the standard for commercial-scale production. Highly acidic foods in plastic pouches or containers require specific migration testing for the acidic food simulant (acetic acid, 3%), which accelerates extraction of some substances. Always test acidic foods specifically, even if you have passed testing for neutral foods.
My supplier gave me a Certificate of Analysis. Is that the same as a Declaration of Compliance?
No, and this confusion costs brands significantly. A Certificate of Analysis (CoA) confirms the physical and chemical properties of a specific production batch. A Declaration of Compliance is a legal document confirming that the material meets food contact regulatory requirements. You need both, but they serve different purposes. A CoA without a DoC does not satisfy food contact compliance documentation requirements.




